Introduction: Cardiovascular complications are a leading cause of early mortality in patients with sickle cell disease (SCD). Chronic anemia, iron overload and repeated microvascular ischemic insults with reperfusion injury often result in progressive myocardial injury and maladaptive remodeling in SCD patients. Patients with SCD develop diffuse myocardial fibrosis, which can be detected by quantifying the myocardial extracellular volume (ECV) using cardiovascular magnetic resonance (CMR) imaging. Our group and others have previously shown that diffuse myocardial fibrosis is highly prevalent in both children and young adults with SCD, including those who have normal ventricular size and function by echocardiography. Additionally, we have shown that the disease modifying therapies (DMT) such as hydroxyurea or chronic transfusions do not appear to be protective against development of diffuse myocardial fibrosis. In a retrospective study, we found no difference in the myocardial ECV in patients who began DMT early (less than 6 years of age) versus later in life (at least 6 years or after) (Morin CE and Sharma A et al. Blood 2023.). Hematopoietic cell transplantation (HCT) can cure SCD, but myocardial effects of the therapy are unknown. Our goal was to evaluate cardiovascular parameters of function and injury, especially the progression of diffuse myocardial fibrosis, by comparing serial CMR studies performed before and after HCT in patients with SCD.
Methods: We performed prospective CMR evaluation of children and young adults with SCD who received a matched sibling donor (MSD) or haploidentical donor (HAPLO) HCT following a reduced toxicity conditioning on a clinical trial (NCT04362293). CMR was performed pre-HCT, and at 1 month, and 12 months post-HCT. CMR-derived ventricular and atrial volumes were calculated. ECV was measured from T1 maps with a modified Look-Locker inversion recovery (MOLLI) sequence in short-axis. T2 and T2* maps were obtained. Regions of interest in the mid-ventricular septum were identified for analysis. We report descriptive statistics for CMR measures along with paired t-tests comparing patient ECV levels at 1-month and 12-months post-HCT to their respective baseline ECV.
Results: Our study cohort consisted of 14 patients with SCD who underwent HCT and had serial CMR evaluations available. Thirteen patients had HbSS genotype and one had HbSC genotype. All patients were receiving DMT, either with hydroxyurea or chronic transfusions prior to HCT. Participants were majority male (57%) with median age of 15.2 years (range 6.8-21.3) at the pre-HCT CMR. Six received an MSD HCT and 8 received a HAPLO HCT.
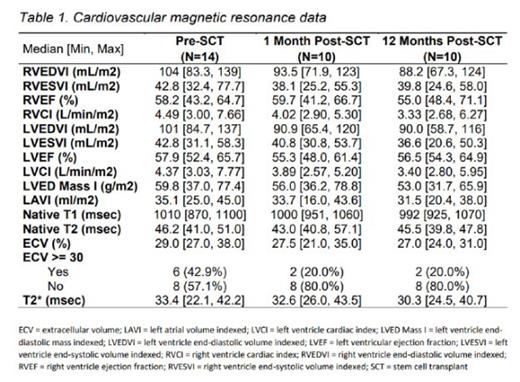
Ten patients each had follow up CMR evaluations performed at 1 month, and 12 months post-HCT. At 1 and 12 months post-HCT ejection fraction was preserved while biventricular volume and cardiac index decreased (Table 1). Compared to pre-HCT, ECV decreased from median 29% [range 27-38] to 28% (range 21-35, P= 0.061) at 1 month and 27% (range 24-31, P= 0.043) at 12 months post-HCT. There was no difference in the cardiac chamber sizes or the ECV measurements of those who received a MSD versus HAPLO HCT.
Conclusions: We observed an improvement in cardiac chamber sizes with preservation of cardiac function as early as one month after HCT in patients with SCD. We further noted a reduction in ECV indicating a decrease in diffuse myocardial fibrosis in these young patients with SCD. To our knowledge, this is the first study to show that diffuse myocardial fibrosis can improve in patients with SCD after HCT. In addition to further corroborating the organ function protective effects of HCT in patients with SCD, these data further demonstrate the use of CMR as an essential non-invasive tool to assess the effects of therapeutic interventions on SCD-cardiac injury.
Disclosures
Sharma:RCI BMT/NMDP: Honoraria, Other: Clinical Trial Medical Monitor; CRISPR Therapeutics: Other: Clinical Trial Site PI, Research Funding; Editas Medicine: Consultancy; Sangamo Therapeutics: Consultancy; Vertex Pharmaceuticals: Consultancy, Other: Clinical Trial Site PI; Medexus Inc: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal